Paul Anastas ( also called the father of green chemistry) and John Warner designs twelve principles of GREEN CHEMISTRY to save the environment.
GREEN CHEMISTRY
It is defined by the Environmental Protection Agency (EPA) as “ The utilization of a set of principles that reduces or eliminates the use or generation of a hazardous substance in the design, manufacture, and application of chemical products.”
TWELVE PRINCIPLES OF GREEN CHEMISTRY
The twelve principles of GREEN CHEMISTRY proposed by Paul Anastas and John are given below,
1. Prevention: It is the first principle of the twelve principles of green chemistry. It is better to prevent waste than treat or clean up waste after it is formed.
2. Atom Economy: – The design of the synthesis of a product should be such that all the materials used in the process is incorporated into the product to the maximum extent.
3. Non Hazardous chemical: – The synthetic methods should be designed, whenever practicable to use and generate substances having little or no toxicity to human health and the environment.
Explanation: –a. The waste products pollute land, water & air e.g. oxides of sulphur & nitrogen produce acid rain. Thus the approach is to prevent the generation of such acidic products which cause acid rain. Therefore, it is better to prevent the formation of waste rather than find methods to clean up these products later.
b. Most of the reactions produce by-products. Thus all the atoms of reactants aren’t utilized in the product. These unutilized substances generate waste. Hence process becomes uneconomical.
c. The materials used should be non-toxic or less toxic. For e.g. processes involving pyridine & p- Naphthyl amine should be avoided as they are carcinogenic (cancer-causing).
Earlier the objective of a chemical reaction was to produce a particular product without bothering about bi-products formed which are wasted. The basic principle of green chemistry is to prevent and minimize the use of hazardous chemicals in the manufacturing process. Therefore, all the chemical processes are re-examined and new processes and products are redesigned such that the wastage of chemicals and pollution is prevented.
Similarly, the manufacture of pesticides using methyl isocyanate is avoided as it is poisonous.
4. Safer Chemicals: – The chemical products should be designed to preserve the efficacy of the desired function while reducing toxicity.
Explanation: – If a drug molecule is toxic then its structure is modified keeping the basic feature so that its function is maintained whereas the toxicity is reduced. Similarly, pesticides like DDT & Gamaxane are toxic in nature & hence they are replaced by phosphate & carbonate pesticides. Currently, bio-pesticides are replacing chemical pesticides.
5. Auxiliary Substances: – The use of auxiliary substances like solvents, separating agents, etc should be made unnecessary whenever possible & innocuous when used.
(Explanation: – The common auxiliary chemical is solvent which becomes toxic waste & also may be inflammable. The dry cleaning solvent i.e. Carbon tetrachloride is found to be toxic, so currently the use of liquid CO2 is used for dry cleaning purposes.)
6. Energy Efficiency: – Energy efficiency is also an important term amongst the twelve principles of green chemistry. The energy requirements of chemical processes should be minimized considering their environmental & economic impacts. The synthetic methods should be carried out at ambient (neither high nor low) temperature & pressure, wherever possible.
Explanation: – The use of fossil fuels & petroleum products as energy sources causes pollutants. Therefore, low-energy processes should be developed. For e.g. fermentation is developed by biotechnology which has minimum energy requirements. Further, microwaves, Ultrasound & solar energy are to be used.
7. Renewable Feedstock: – The raw material should be renewable rather than depleting whenever technically & economically feasible.
Explanation: – Coal & petroleum as sources of raw materials are exhaustible sources. Therefore, materials such as bio-mass (plant or animal product) is used which are renewable & harmless. For e.g. adipic acid is currently prepared from glucose which is obtained from corn starch.
8. Derivatization: During synthesis, unnecessary derivatization such as blocking group, protection, or deprotection, or temporary modification of physical & chemical; processes should be avoided whenever possible.
The derivatization increases the no. of steps which further increases the wastage & energy consumption. Therefore, the processes with a minimum no. of steps are being explored. The “Single ⌯ Plot” reactions are the best option.
Out of the twelve principles of green chemistry, we also think about the unnecessary preparation of derivatives/intermediates.
9. Catalysis: – The catalytic reagents which are selective are superior to stoichiometric reagents. The catalysts & bio-catalysts are used to carry out the reaction. It uses less time & energy & the catalyst can be recovered. The usual reagents generate waste products. Hence it is also a valuable point amongst the twelve principles of green chemistry.
10. Degradable Products: – Chemical products should be designed such that at the end of their function, they break down into insignificant degradation products.
The products which are used should finally degrade into harmless products. For e.g. DDT isn’t bio-degradable. Hence causes environmental pollution. Similarly, plastics are also not bio-degradable therefore, plastics containing cellulose are being prepared to get bio-degradable plastics.
11. New Analytical Methods: -New analytical methods have to be developed to allow online process monitoring & control prior to the formation of hazardous substances.
The conditions of the reaction should be continuously monitored to check the formation of toxic material. For e.g. in the preparation of ethylene glycol if the temperature goes up it generates a highly toxic substance called dioxin. Therefore, the temp should be properly controlled to prevent its formation.
12. Accident Prevention: – It is one of the important principles among the 12 principles of green chemistry. The substances & their form used in the chemical process should be chosen to minimize the potential for chemical releases, accidents, explosions & fires.
- Inflammable liquids like benzene, ether, sulphur & carbon disulfide may cause a fire hazard.
- During the nitration of toluene to prepare INT proper conditions should be maintained to prevent an explosion.
- During the high-pressure reactions of chemicals should be controlled.
For e.g. Release of methyl isocyanate (MIC) by Union Carbides caused a lot of loss of life.
I hope you understand the principles of green chemistry.
Atom Economy (or Atom Efficiency):
It is a measure of the efficiency with which the atoms of reactants in any are incorporated into the desired product. It is expressed as a percentage, which is given by.
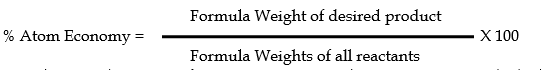
In accordance with the concept of atom economy, a good reaction is one in which all the atoms in the reactant appear in the final product. ( No waste at all)
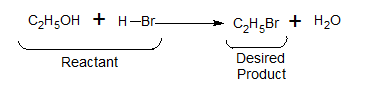
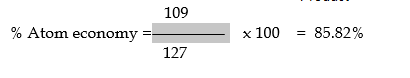
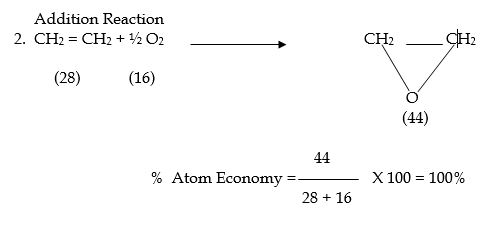
Note: –
- Addition & rearrangement reactions have the highest atom economy whereas substitution & elimination have lower values.
Reaction having 80% or more atom economy should be carried obtain, rest i.e. below should be avoided.
E-factor ( Effluent factor): –
It is called the Effluent factor which is the measure of the amount of waste products generated during the process.
The e-factor is the ratio of wastage produced in kgs to the weight of desired product in kg.
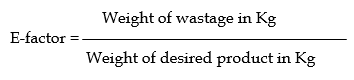
If the atom Economy in a reaction is low, wastage will be high & this increases the E-factor and such a reaction shouldn’t be used. E-factor also depends on the size of the manufacturing facility. For bulk production the E- factor should be very low because the disposable waste should be minimum.
Green Chemistry: –
Practicing chemistry in a manner that is environmentally friendly is the essence of green chemistry. The environment protection agency [EPA] defines Green Chemistry as “The Utilization of a set of principles that reduces or eliminated the use or generation of the hazardous substance in the design, manufacture & application of chemical products”.
The term green chemistry as adopted by the IUPAC working party on Synthetic pathways and process is
“The invention, design, and application of chemical products and processes to reduce or to eliminate the use and generation of hazardous substance”.
Please also read..
Q: Who gave twelve principles of green chemistry?
Paul Anastas (also called the father of green chemistry) and John Warner designs the twelve principles of Green Chemistry in 1991.
Q: Who is called the father of green chemistry?
Paul Anastas is known as the Father of Green Chemistry. He designs twelve principles of Green Chemistry in 1991.
Q: Why is Paul Anastas known as the Father of Green Chemistry?
Paul Anastas is known as the Father of Green Chemistry because of his great contribution to Green Chemistry for the survival of planet earth.
Q: What is Green chemistry?
Green Chemistry is the designing of a process or route of chemical reaction/process that reduces or eliminates the generation of hazardous waste. Please refer twelve principles of Green Chemistry for more details.
Thanks a lot for the helpful posting. It is also my belief that mesothelioma cancer has an really long latency period of time, which means that warning signs of the disease may well not emerge till 30 to 50 years after the primary exposure to asbestos. Pleural mesothelioma, which is the most common style and affects the area within the lungs, will cause shortness of breath, upper body pains, and a persistent coughing, which may lead to coughing up body.
Thanks for your posting. One other thing is when you are advertising your property alone, one of the concerns you need to be aware of upfront is when to deal with property inspection accounts. As a FSBO owner, the key concerning successfully transferring your property in addition to saving money in real estate agent revenue is information. The more you are aware of, the more stable your property sales effort might be. One area that this is particularly essential is information about home inspections.
Can I just say what a aid to find somebody who truly knows what theyre speaking about on the internet. You definitely know find out how to carry an issue to mild and make it important. Extra people must learn this and understand this aspect of the story. I cant consider youre not more well-liked because you undoubtedly have the gift.
Heya just wanted to give you a quick heads up and let you know a few of the images aren’t loading properly. I’m not sure why but I think its a linking issue. I’ve tried it in two different web browsers and both show the same outcome.
Please let me know if you’re looking for a article author for your weblog. You have some really great articles and I think I would be a good asset. If you ever want to take some of the load off, I’d absolutely love to write some content for your blog in exchange for a link back to mine. Please send me an e-mail if interested. Regards!
This website was… how do I say it? Relevant!! Finally I have found something that helped me. Thanks!
I was wondering if you ever thought of changing the structure of your blog? Its very well written; I love what youve got to say. But maybe you could a little more in the way of content so people could connect with it better. Youve got an awful lot of text for only having one or two pictures. Maybe you could space it out better?
Thank you dear i will improve it
I have observed that car insurance businesses know the motors which are prone to accidents as well as other risks. Additionally , they know what kind of cars are prone to higher risk as well as higher risk they may have the higher the premium fee. Understanding the basic basics of car insurance just might help you choose the right type of insurance policy that will take care of the needs you have in case you happen to be involved in an accident. Thanks for sharing your ideas on the blog.
Thanks for making me to attain new suggestions about computer systems. I also hold the belief that one of the best ways to help keep your notebook computer in best condition is a hard plastic case, or maybe shell, that fits over the top of your computer. Most of these protective gear tend to be model distinct since they are made to fit perfectly over the natural covering. You can buy them directly from the owner, or via third party places if they are intended for your mobile computer, however its not all laptop will have a spend on the market. All over again, thanks for your points.
Hi there would you mind stating which blog platform you’re working with?
I’m looking to start my own blog in the near future but I’m having
a hard time selecting between BlogEngine/Wordpress/B2evolution and
Drupal. The reason I ask is because your design and style seems different then most blogs and I’m looking for something unique.
P.S My apologies for getting off-topic but I had to ask!
I am always searching online for tips that can aid me. Thx!
I have recently started a blog, the info you offer on this website has helped me tremendously. Thanks for all of your time & work.
thank you dear
Wow, incredible blog structure! How long have you ever been running a blog for? you made running a blog look easy. The entire glance of your web site is great, as well as the content!
thank you dear
I conceive this site contains some rattling good information for everyone. “It is easy enough to define what the Commonwealth is not. Indeed this is quite a popular pastime.” by Elizabeth II.
I like the valuable information you supply for your articles. I will bookmark your blog and take a look at again right here frequently. I am rather certain I’ll be told many new stuff proper right here! Good luck for the next!
thank you dear
There’s certainly a great deal to learn about this issue. I really like all of the points you made.
Thank you very much